Participant information sheet (Clinical trial) (BEPKO3) – v3 (20-11-25)
Title of Project: BEhaviour change for People with clinically diagnosed Knee Osteoarthritis: Clinical trial (BEPKO-3)
Name of researcher: Stephen Preece
You are invited to take part in a research study to help us develop and test a new treatment for people who live with knee osteoarthritis. Before you decide, it is important for you to understand why the research is being done and what it will involve. This document gives you important information about the purpose, benefits and potential disadvantages related to participating in the study. Please take time to read the following information carefully. If you have any questions, then feel free to contact the researcher whose details are given at the end of the document. We recommend taking 24 hours to decide whether you wish to take part. Your decision whether to take part in the study or not will have no bearing on your medical care.
What is the purpose of the study?
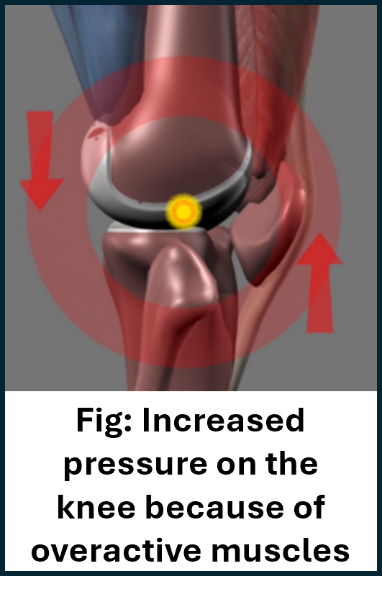
We want to understand whether a new form of physiotherapy could be effective for people with knee osteoarthritis who have not benefited from current physiotherapy treatment. This new form of physiotherapy aims to teach patients how to stand and move with less muscle tension in their knees and throughout their body. It also teaches patients to change the way they think about and react to pain. Muscle biofeedback is used throughout the treatment to allow patients to directly observe, on a computer screen, when their muscles are tense or relaxed. The idea behind the treatment is to reduce muscle tension, lower the pressure on the knee and reduce knee pain. This new treatment is very different from existing physiotherapy (which you will have tried before) which focuses on muscle strengthening exercises.
Why have I been invited to take part?
You have been invited because you have a diagnosis of knee osteoarthritis. If you are still experiencing pain despite trying existing physiotherapy, then you may benefit from this new treatment. We have invited NHS patients with a diagnosis of knee osteoarthritis, along with NHS physiotherapists who have been trained to deliver this new treatment, to take part in this study.
Do I have to take part?
No, taking part is completely voluntary. If you are interested, contact the researcher (details at the end of this information sheet).
What will happen to me if I participate in this study?
If you are interested in taking part in this study a member of the research team will perform an eligibility check. If you are eligible, you will sign a consent form and will first be asked to complete a set of questionnaires which allow us to understand your symptoms and how your knee osteoarthritis interferes with your daily life. You will also be asked to complete a set of questionnaires which allow us to understand how often you access healthcare services (e.g. GP visits) and how your condition affects your capacity to work. You will be required to complete these same questionnaires on two other occasions: 6 months and again at 12 months after you enrol on the study.
When you complete the first set of questionnaires, you will also complete a form which will give us permission to look at any previous x-ray data from your knee. We will also ask your permission to access your NHS data on knee surgery that you might choose to have during a 10-year period after the study is completed. Once we receive the first set of questionnaires, we will write to your GP to explain that you are taking part in this study. You will then be randomly allocated (like flipping a coin) to either receive the new treatment (group 1) or to be in the control group (group 2).
If you are in Group 1:
You will be asked to continue to access NHS care for your knee osteoarthritis as you would if you were not in the trial. In addition, you will receive the new physiotherapy treatment across seven weekly sessions, each lasting 1 hour. At the start of the treatment, the physiotherapist will explain how reacting and thinking differently about your osteoarthritis has the potential to reduce pain. You will then be taught how to consciously relax your knee muscles and how to relax your stomach muscles using a special breathing technique. The next stage of the treatment is focused on teaching you to stand with less muscle tension. This is achieved using simple exercises which enable you to build awareness of patterns of muscle tension, particularly around your knees. Once you can stand with relaxed knees, the focus shifts to movement. Muscle biofeedback is used to visualise your muscle patterns on a screen. You are then guided through a process in which you learn to perform daily movements with less muscle tension.
As you progress through the treatment, you will gain a new experience of standing and moving which may feel strange at first but which you will get used to. To help you understand many of the ideas which underlie the new treatment, animated instructional videos are used. These videos are watched on a tablet or laptop computer. If you don’t have access to a computer, we will provide you with a paper booklet. The aim is to deliver weekly sessions of the treatment. However, we will offer flexibility on appointment times to ensure you can complete the full treatment. Note that, if you do decide to withdraw, you would no longer receive the treatment. If you want to withdraw, please notify the study representative whose contact details are provided at the end
We want to understand how good physiotherapists are at delivering the new treatment. To do this, we need to either have another physiotherapist observe the treatment and/or video some of the treatment sessions. Any video footage will be stored in a secure folder on the University of Salford network, will only be accessible to the research team and will be permanently deleted after it has been reviewed (typically within two weeks). However, if you don’t want to be videoed, then you can refuse this.
After you have received the treatment, we will offer you the opportunity to be interviewed about your experiences of being involved in the study and to understand what you think about the new treatment. These interviews will be carried out over the phone or via video conference and will be an informal way of expressing your opinions. We will record these interviews, but all data will be completely anonymised and only the research team (not the physiotherapist) will have access to your anonymised opinions. The interview recordings will be stored in a secure folder on the University of Salford network. The interviews will be transcribed (typed onto a computer) by the research team. After this transcription, the recording will be permanently deleted, this will typically be within two weeks of the recording.
If you are in Group 2:
You will be asked to continue to access NHS care for your knee osteoarthritis as you would if you were not in the trial.
Expenses and payments?
Unfortunately, we are unable to pay you to receive the new treatment or cover any associated travel expenses. However, you will receive £20 when you are invited to complete the questionnaires sent to you 6 months and 12 months after enrolling on to the study. You will also receive £20 if you agree to be interviewed after receiving the treatment.
What are the possible disadvantages and risks of taking part?
This is a very simple, straight forward study. The physiotherapist will be using techniques which are used in routine clinical practice, and which do not carry any risk.
What are the possible benefits of taking part?
If you are in group 1 you will receive 7 sessions of the new physiotherapy treatment which may reduce your knee pain. If you are in group 2 then you will continue to access your NHS care as normal. Being part of this trial will allow the research team to collect valuable scientific data which will inform the NHS on how best to provide care for people who live with knee osteoarthritis.
Who is organizing and funding the research?
This study is being led (and sponsored) by the University of Salford. The study has been funded by the National Institute for Health Research, which is part of the NHS.
How will we use information about you?
We will need to use information from you for this research project. This information will include your initials, NHS number, knee x-ray, name and contact details. People will use this information to do the research or to check your records to make sure that the research is being done properly. People who do not need to know who you are will not be able to see your name or contact details. Your data will have a code number instead.The University of Salfordis responsible for looking after your information. We will share your information related to this research project with other universities and NHS organisations. We will keep all information about you safe and secure by ensuring it is saved on password protected computers within encrypted files.
International transfers
We may share or provide access to data about you outside the UK for research-related purposes to enable other researchers to compare our findings with other research into osteoarthritis. However, we will not share any personal information, and we will ensure that you can’t be identified from the data.
How will we use information about you after the study ends?
Once we have finished the study, we will keep some of the data so we can check the results. We will write our reports in a way that no-one can work out that you took part in the study. We will keep your study data for 10 years after the study completes. The study data will then be fully anonymised and securely archived or destroyed. We are happy to send each participant in the study a summary of the results. Please indicate on the consent form if you would like to receive this summary and confirm that you are happy for us to retain your contact information for 1 year after the study is completed to allow us to send this information to you. If you take part in an interview, then we may use anonymised quotes when writing up our results. No identifiable data will be kept after the end of the study (apart from contact details if you would like a summary of the results).
What are your choices about how your information is used?
You can stop being part of the study at any time, without giving a reason, but we will keep information about you that we already have. You have the right to ask us to access, remove, change or delete data we hold about you for the purposes of the study. You can also object to our processing of your data. We might not always be able to do this if it means we cannot use your data to do the research. If so, we will tell you why we cannot do this.
Where can you find out more about how your information is used?
You can find out more about how we use your information at https://www.salford.ac.uk/privacy, by asking one of the research team or by contacting the Data Protection Officer at the University of Salford at foi@salford.ac.uk.
What if there is a problem?
The university has insurance to cover against any harm to you which may occur whilst you are taking part in these tests. However, if you decide to take legal action, you may have to pay for this. If you wish to complain, or have any concerns about any aspect of the way you have been approached or treated during the course of this study, you can contact the project supervisor Prof Stephen Preece (email: s.preece@salford.ac.uk) and if you are not happy you may then contact Dr Katy Szczepura, Ethics Chair, Allerton Building, University of Salford, M5 4WT via email: K.Szczepura@salford.ac.uk.
Further information and contact details:
We appreciate your interest in this study and hope to welcome you at the School of Health and Society, University of Salford. If you are interested in taking part in this study then please get in touch. Contact details and the online form can be found on our BEPKO-3 website.